O Orbital Diagram
Orbitals electron clagett orbital ic chemistry organic Molecular chemistry orbitals orbital diagram energy bonding level edu wave two h2 theory atomic chemwiki molecule bond libretexts delocalized function Electron orbitals chemistry quantum electrons numbers model electronic structure arrangement introductory orbital number energy atoms chem level libretexts atomic chapter
Pre-Chemistry
Orbitals orbital quantum atomic electrons chem describe Atomic orbitals Orbits electrons distribution electron shell teachoo nucleus
Orbital diagram example 2s 1s 2p
Orbital molecular diagrams ru2 stateMolecular orbital theory Ch 1 : electrons and orbitalsElectron configuration.
4.9: molecular orbitalsOrbital molecular li2 be2 orbitals molecule atomic electrons wiring 9.7: molecular orbitalsDiagram molecular orbital f2 mo theory practice chem model hybridization shown problem below.
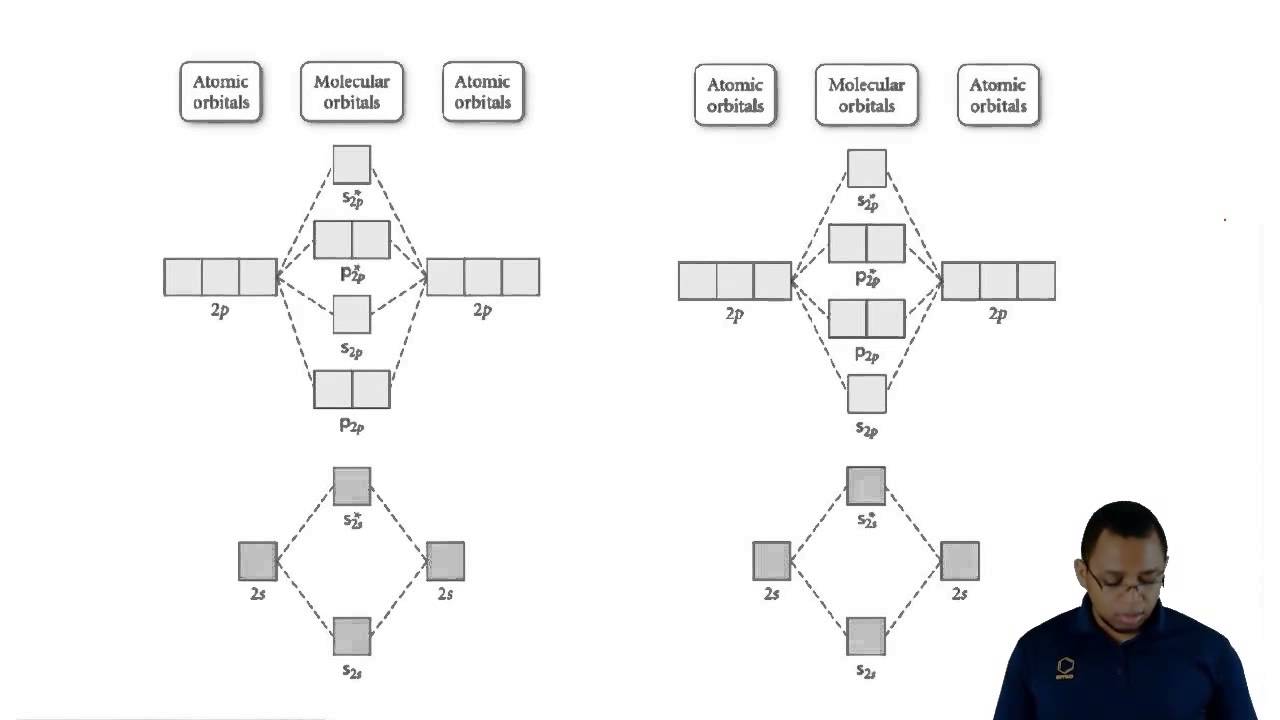
Empty orbitals in molecular orbital theory?
Ch 1 : electrons and orbitalsMolecular orbital diagram diatomic molecules theory orbitals bonding cl2 diagrams energy bond electron chemistry level delocalized second homonuclear period row Orbitals molecular atomic orbital educator diagrams configuration electronDrawing atomic and molecular orbitals diagrams for molecules.
Orbital configuration atomic electronOrbital overview sulfur caroline monahan Orbital diagram electron configuration chart electrons atom level sublevel atomic structure circle most there each representation wikimedia commons ccOrbital diagrams — overview & examples.

Molecular orbitals bonding diatomic orbital atomic pi star chemistry theory delocalized molecules np atoms bond libretexts chem formation figure geometry
Electron configuration chart6.6: 3d representation of orbitals The art of organic chemistry: 2: atomic and molecular orbitalsOrbitals shape atom orbital atomic nodes representation hydrogen electron angular energies chem libretexts orbitaal molecular equivalent bonding atoms wisc unizin.
Chapter 6.5 delocalized bonding and molecular orbitals3.7: electron arrangement- the quantum model Orbitals shape shapes chemistryMolecular orbital theory.

Orbital orbitals electron atoms science chemistry britannica
Molecular orbital theoryOrbital quantum numbers orbitals 2p 2s 1s atom electrons chem carbon describe set familiar meaning should try questions some ucalgary Chemistry online: shape of orbitals....Distribution of electrons in different orbits [with examples].
Understanding molecular orbital theoryOrbital atoms orbitals quantum radon grandinetti Orbital diagramElectron orbital.

Orbital molecular theory
Molecular orbitals atomic orbital molecules socratic mo laidOrbital molecular he2 theory o2 be2 bonding paramagnetic diamagnetic mo orbitals electrons diatomic molecules chem unpaired antibonding atoms inorganic mixing High school chemistry/shapes of atomic orbitalsOrbital molecular diagrams origins molecules mathematics does.
Chm 501 lecture 7 introduction to molecular orbital theoryMolecular orbital diagram atomic orbital electron configuration, png Orbitals shapes atomic chemistry probability patterns figureOrbitals shapes quantum theory orbital diagram figure sublevel chemistry electrons atoms axis sublevels chem orientation spherical their magnetic number shaped.

Development of quantum theory
Molecular orbitals electrons bonding libretexts valence chem delocalizedChm orbitals theory molecular orbital .
.





